Dalton’s atomic theory states the following: Matter is composed of exceedingly small particles called atoms. Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). Modern understandings of chemistry can be traced back to John Dalton who, in 1807, first published his hypothesis that the behavior of matter could be explained using an atomic theory. Best Known For: Chemist John Dalton is credited with pioneering modern atomic theory. Isobars are atoms that have the same characteristics in different. Isotopes are atoms of the same element that exhibit distinct properties.
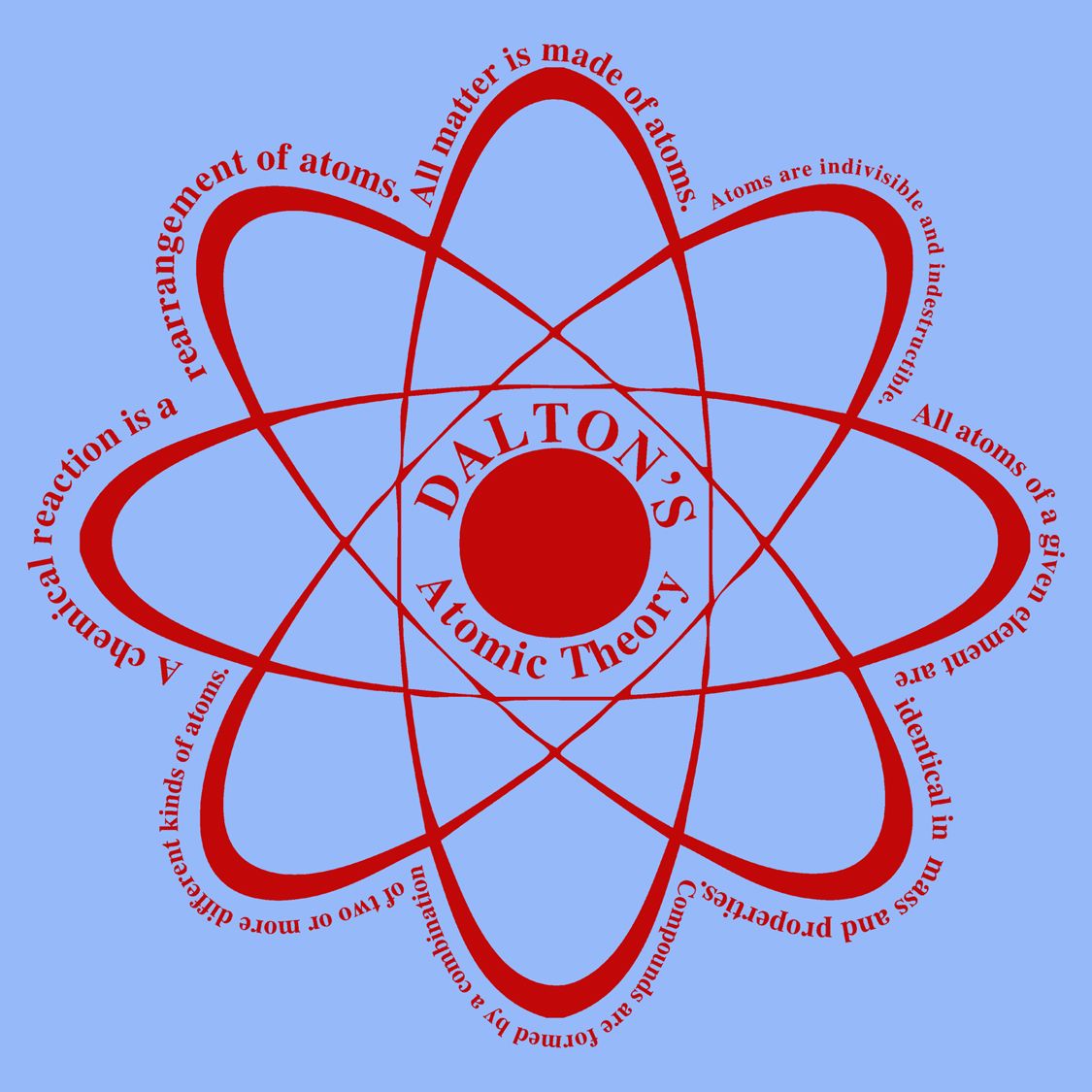
Atoms of a given element are identical in size, mass, and other properties. Atoms cannot be subdivided, created, or destroyed.
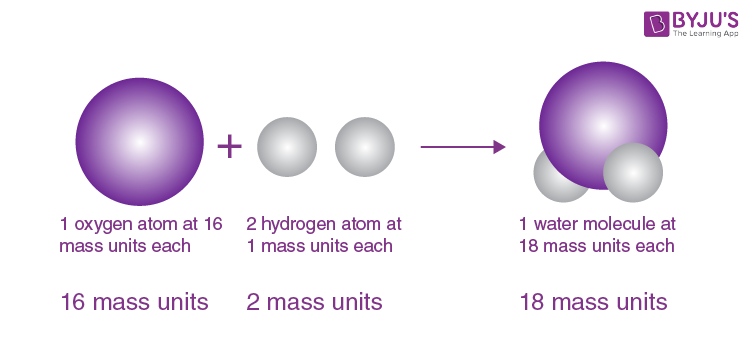
The general tenets of this theory are: All matter is composed of extremely small particles called atoms. See all videos for this article By far Dalton’s most influential work in chemistry was his atomic theory. The contradiction of modern atomic theory with daltons atomic theory: Atoms are divided into protons, neutrons, and electrons, according to modern atomic theory, which contradicts Daltons atomic theory. Dalton proposed his atomic theory in 1804. However, Dalton failed to recognize natural tendencies of elements in nature. Atomic theory of John Dalton See how John Dalton built his atomic theory on principles laid out by Henry Cavendish and Joseph-Louis Proust John Dalton and the development of the atomic theory. Daltons modern atomic theory, proposed around 1803, is a fundamental concept that states that all elements are composed of atoms. Dalton proposed a list of atomic weights in 1805. Daltons empirical, experimentally-based work marked the first scientific theory of the atom. Please read on the concept of matter here. Recall that chemistry is the study of matter and the changes they undergo. The Law of Definite Proportions applies when elements are reacted together to form the same product. Atomic theory states that matter is composed of discrete units, called atoms. Dalton’s Atomic Theory: John Dalton, an English chemist is considered the father of modern chemistry because of the understanding he brought into the composition of matter and the basis for their chemical reactions. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C.

This 2-to-1 ratio means that the brown compound has twice the amount of chlorine per amount of copper as the green compound.\): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B. Dalton The theory of the atom would remain unproven until 1803, when an English scientist presented the proof. doesn’t violate the law of multiple proportions, the law of conservation of mass, and the law of constant proportions.

